Exploring the diffusion behavior of urea aqueous solution in the viscose film by ATR-FTIR spectroscopy
Citation
Yan Dong, Lei Hou*, and Peiyi Wu*. Exploring the diffusion behavior of urea aqueous solution in the viscose film by ATR-FTIR spectroscopy. Cellulose 2020, 27, 2403-2415.
Abstract
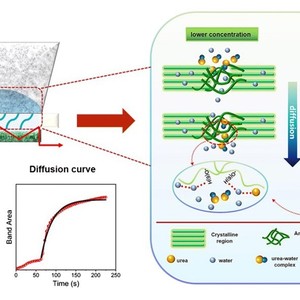
The diffusion mechanism of the urea aqueous solution in the viscose film is investigated by time-resolved attenuated total reflectance-Fourier transform infrared spectroscopy combining with perturbation correlation moving window technique. Two stages can be observed during the whole diffusion process: (1) water diffuses from urea aqueous solution into the viscose film, (2) urea and water diffuse simultaneously through the viscose film. Additionally, the diffusion curve of urea in the viscose film fits Fickian diffusion model, and the diffusion coefficients can be calculated accordingly. It is shown that the diffusion rate of urea slightly decreases with increasing concentration. Furthermore, considering the frequency shift of bands attributed to C–O stretching vibration (mainly –CH2–O(6)H groups) of cellulose, it is suggested that water molecules are the dominant component interacting with cellulose by first breaking the native hydrogen-bonding network in the amorphous region and then building new cellulose-water hydrogen bonds during the diffusion process. On the other hand, urea molecules merely decrease the diffusion mobility of aqueous solutions by immobilizing the water molecules and forming urea-water associations in the viscose film. Thus, it is inferred that urea molecules play an indirect role by influencing the mobility of water molecules rather than directly interacting with cellulose chain in the viscose film during diffusion. The results herein could provide guidance in the filed of urea’s function in reactive dye printing and cellulose dissolution.